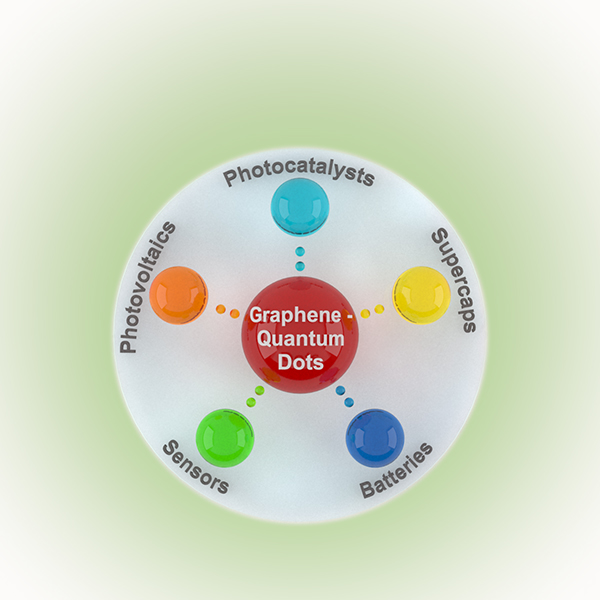
The renewable sources of energy (photovoltaics, wind and water turbines) are ecologically benign but they have a major drawback, i.e. the dependence of cyclic (day/night) and climatic (clouds, wind current and sea waves) factors for the production of electricity. Here comes a back-up solution to solve this issue such as a system of battery and supercapacitors (supercap). The surplus electricity produced by the respective device during the day or windy period is stored in battery/supercapacitor and it is delivered to the power grid when necessary (night, clouds). Moreover, there is a significant demand increase of energy for portable devices and especially for transportation, aiming to reduce the greenhouse gases in the atmosphere.
A quick twofold response by improving the existing and developing of new technologies in the energy field for overcoming above mentioned issues is needed. The nature and form of the novel put-in materials could be the base for the both approaches.